Abstract
Background: Myelodysplastic Syndromes (MDS) are genetically and hematologically diverse stem cell malignancies pathogenetically linked to constitutive innate immune activation. Other than rare germline mutations and age, the only known predispositions to adult MDS include prior cytotoxic therapy, clonal hematopoiesis, and autoimmune or chronic inflammatory disorders. Few studies investigating the genetic susceptibility to MDS have been performed owing to the limitations of SNP-array sample size. Here, we report the results from the first unbiased genome wide analysis of germline polymorphisms associated with non-del(5q) MDS using a multinational curated data set.
Methods: Association analyses were performed on 2 sample sets (set 1: 555 cases, 2,964 controls; set 2: 352 cases, 2,640 controls) and combined by meta-analysis. Standard SNP- and sample-level QC was applied. Haplotype reference consortium (HRC) imputation was done by the Michigan imputation server (Rsq>0.4) providing 23,278,269 markers for analysis. Gene expression sequencing was performed on an independent sample set from the National Taiwan University Hospital (213 MDS cases, 20 healthy donors; HumanHT-12 v4 Expression BeadChip; Chuang et al. Leukemia 2015). Functional analyses were performed as described.
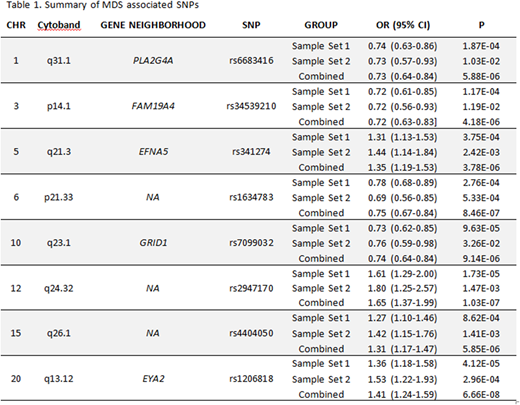
Results: Eight MDS associated loci were identified with lead variants, rs6683416, rs34539210, rs341274, rs1634783, rs7099032, rs2947170, rs4404050, and rs1206818, at 1q31.1 (PLA2G4A), 3p14.1 (FAM19A4), 5q21.3 (EFNA5), 6p21.33, 10q23.1 (GRID1), 12q24.32, 15q26.1, and 20q13.12 (EYA2), respctively. Odds ratio (OR) and p-values of each are listed in Table 1. Using gene expression profiling in an independent MDS sample set, we found expression of these five candidate genes was significantly increased in MDS vs controls (p<0.01). Using the same independent sample set, we found higher PLA2G4A and lower EYA2 expression were associated with reduced overall survival (p=0.039 and p=0.037, respectively). Importantly, EYA2 was an independently favorable risk factor irrespective of age, gender and IPSS-R (RR, 0.665; p=0.048). High PLA2G4A expression was associated with NRAS (p<0.001), RUNX1 (p=0.012), ASXL1 (p=0.007), and EZH2 (p=0.038) somatic mutations whereas higher FAM19A4 expression associated with KRAS mutations (p=0.045). Importantly, PLA2G4A, FAM19A4, GRID1, and EYA2 are involved in the regulation of innate immune signaling. We previously reported that across all somatic gene mutation classes, MDS cases biologically converge upon the redox-sensitive, Nlrp3 inflammasome to drive pyroptosis, a caspase-1 mediated innate immune cell death (Basiorka et al. Blood 2016). PLA2G4A encodes phospholipase A2 (PLA2) that inhibits inflammasome activation. Further, PLA2 is required for MAPK phosphorylation of S100A9, a key danger associated molecular pattern (DAMP) and TLR4 ligand implicated in the induction of pyroptosis in MDS progenitors. FAM19A4 encodes a cytokine whose expression is upregulated in response to endotoxin exposure suggesting a role in immune modulation. EYA members activate innate immune response in part through recognition of cytosolic double-stranded DNA, which can serve as a DAMP triggering inflammasome assembly. To investigate the possible role of EYA2 in inflammasome activation in MDS, we overexpressed EYA2 in the monocytic cell line, THP1. EYA2 overexpression induced caspase-1 and maturation of IL-1β, indicating inflammasome activation. These findings were accompanied by NFκB phosphorylation consistent with innate immune signal priming. We next treated MDS primary bone marrow specimens (n=3) with the EYA2 inhibitor, MLS000544460, demonstrating a significant, dose-dependent improvement in colony recovery consistent with restoration of effective hematopoiesis.
Conclusion: We describe here the first MDS susceptibility loci ever identified the majority of which have a direct relationship to innate immune activation, a driver of MDS pathogenesis. Expression of genes housing these loci is increased in MDS with demonstrable prognostic and biological relevance. Further, functional studies implicate EYA2 as a novel, biologically rational target for MDS treatment. The direct functions of each polymorphism as well as the potential relationship between these predisposition loci to age-related clonal hematopoiesis merits further investigation.
Cluzeau:Amgen: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Jazz Pharma: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Sanofi: Speakers Bureau; AbbVie: Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Menarini: Consultancy; Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Pfizer: Speakers Bureau. Sallman:Celgene: Research Funding, Speakers Bureau. Sokol:Spectrum Pharmaceuticals: Consultancy; Seattle Genetics: Consultancy; Mallinckrodt Pharmaceuticals: Consultancy. Maciejewski:Ra Pharmaceuticals, Inc: Consultancy; Alexion Pharmaceuticals, Inc.: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau; Apellis Pharmaceuticals: Consultancy; Ra Pharmaceuticals, Inc: Consultancy; Apellis Pharmaceuticals: Consultancy; Alexion Pharmaceuticals, Inc.: Consultancy, Membership on an entity's Board of Directors or advisory committees, Speakers Bureau. List:Celgene: Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal